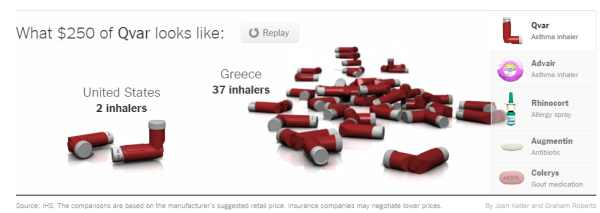
Unlike other countries, where the government directly or indirectly sets an allowed national wholesale price for each drug, the United States leaves prices to market competition among pharmaceutical companies, including generic drug makers. This has resulted in a vicious cycle of repatenting old drugs that use to cost pennies with new delivery systems and processes that now allow the pharmaceutical companies to charge extremely high prices. This article focuses on the skyrocketing prices of asthma medicines.
Questions:
1. Besides repatenting, what other factors are increasing the prices of older drugs in the U.S.?
2. Why are these practices allowed to continue in the U.S.?
3. Take the example of California’s Medicaid program that spent $61 million on asthma medicine last year. Using the information in the article, calculate the cost savings for Medicaid program in California.
(a) Assume that all of this is Rhinocort Aqua, which costs $250 in California, but $7 in Europe.
(b) Assume that all of this is Pulmicort, which costs $175 in the U.S., but $20 in Britain.
(c) Assume that all of this is Albuterol, which costs $100 in the U.S., but cost $15 before repatenting.
4. Discuss the ethics and legality of Pay for Delay schemes.
Source:
Rosenthal, E. (2013). The Soaring Cost of a Simple Breath. The New York Times, Oct. 12 (Retrievable online at http://www.nytimes.com/2013/10/13/us/the-soaring-cost-of-a-simple-breath.html?pagewanted=all&_r=0)